 
Every point is illustrated by case studies, including a commercially available albumin, a small RNA virus isolated from plants, as well as four soluble proteins and a ribonucleoprotein assembly purified and characterized by students in the frame of their master degree. Variables like temperature, solvent viscosity, and inter-particle interactions may also influence particle size determination. To ensure reproducible quantitative data, attention should be paid to controlling the preparation and handling of proteins or assemblies because variations in the state of aggregation, induced by minor changes in experimental condition or technique, might compromise DLS results and affect protein activity. It does this by illuminating the particles with a laser and analysing the intensity fluctuations in the scattered light. It reviews the basic concepts of light scattering measurements and addresses four critical aspects of the analysis and interpretation of DLS results. Dynamic Light Scattering (also known as PCS - Photon Correlation Spectroscopy) measures Brownian motion and relates this to the size of the particles. 
This article is written for graduate and undergraduate students with access to DLS and for faculty members who wish to incorporate DLS into a lab activity, a practical course or research. 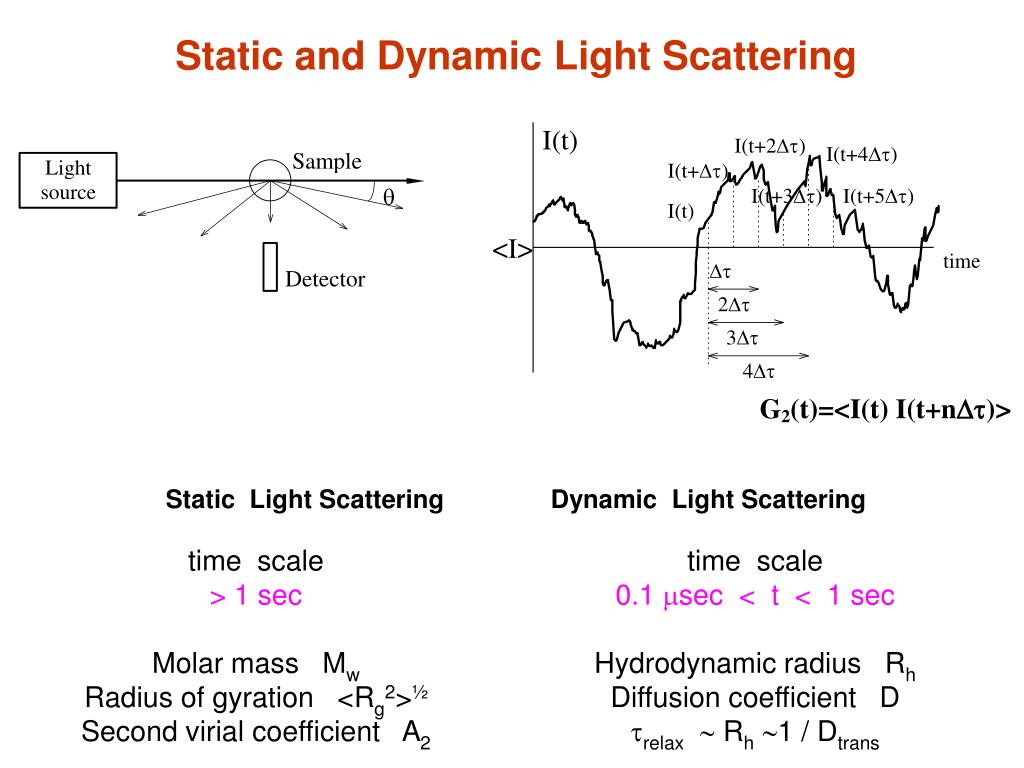
From there the HDD and the polydispersity index (PDI) are calculated. Figure 1: Exemplary correlation function of a DLS measurement. Dynamic light scattering (DLS) analyses are routinely used in biology laboratories to detect aggregates in macromolecular solutions, to determine the size of proteins, nucleic acids, and complexes or to monitor the binding of ligands. Dynamic Light Scattering (DLS) 1, 2, otherwise known as Photon Correlation Spectroscopy (PCS) or Quasi-Elastic Light Scattering (QELS), is a light scattering technique widely used to. Dynamic light scattering (DLS) results DLS measurements provide results as the hydrodynamic diameter (HDD) and peak size (see Figure 1 and 2). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
